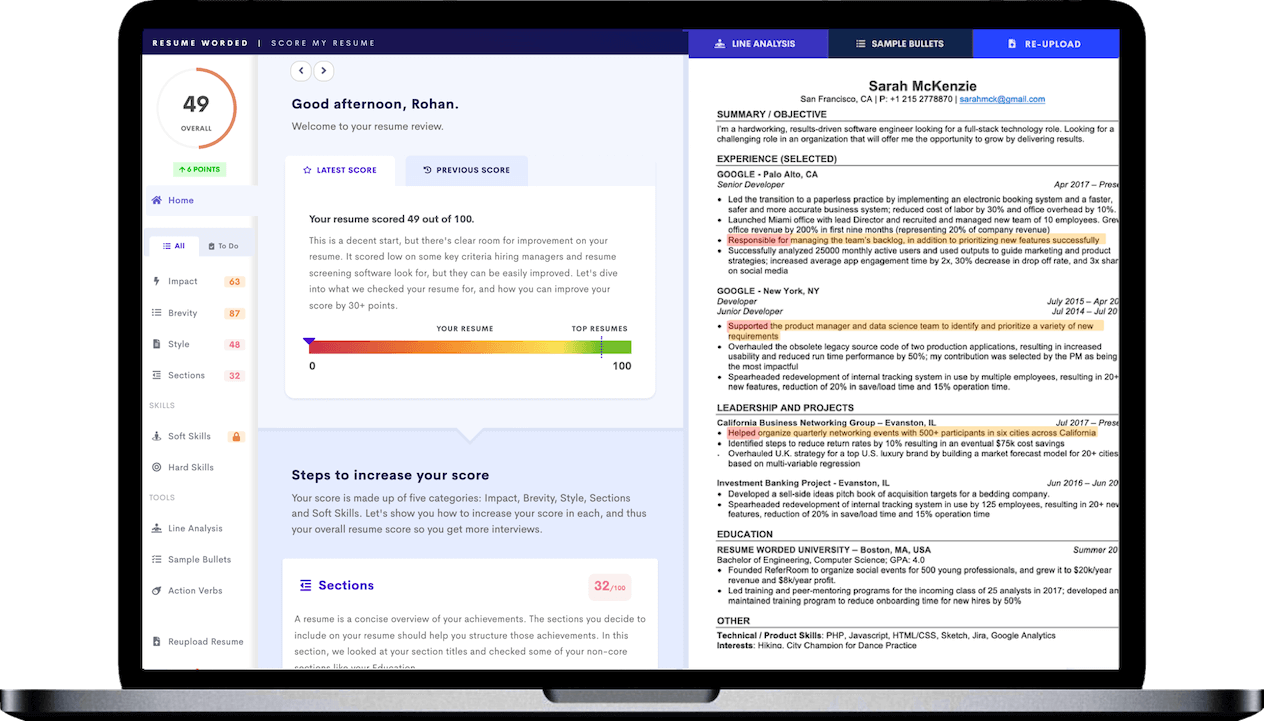
Showing enrollment success
By stating that you 'successfully enrolled 80% of targeted patients' you are demonstrating your effectiveness in patient recruitment. This is a key metric in clinical research, as a higher patient enrollment rate directly translates to more efficient and successful trials.
Demonstrating impact
Sharing that you 'reduced data entry errors by 30%' gives a concrete example of the positive impact you have had. It shows your attention to detail, understanding of data management systems, and the ability to implement improvements to increase efficiency and accuracy.



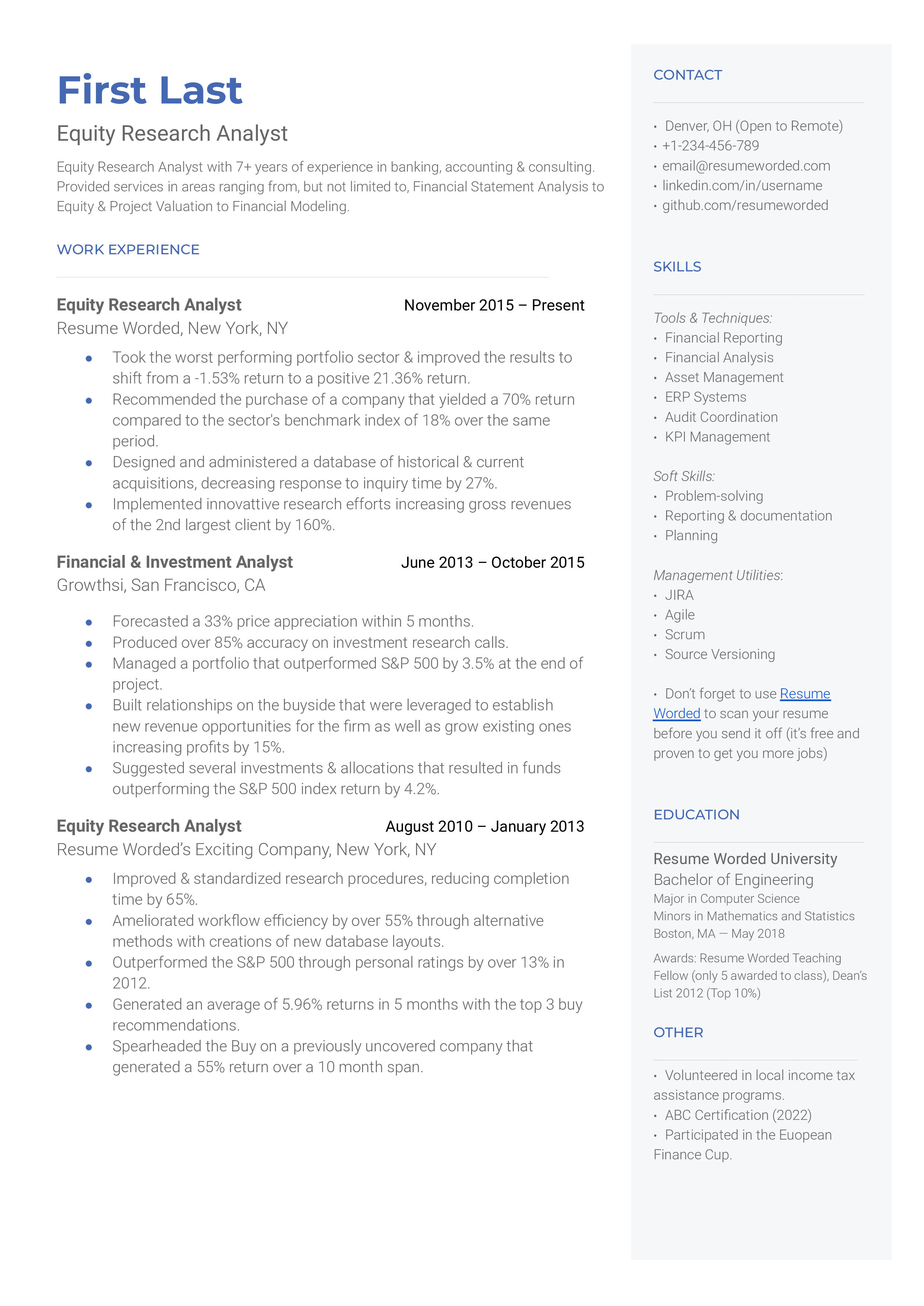













Highlighting experience duration
Specifying the 'decade-long tenure' as a Clinical Researcher immediately establishes your credibility and reliability. It shows that you have maintained and grown in the same field, which speaks volumes about your dedication, perseverance, and expertise.