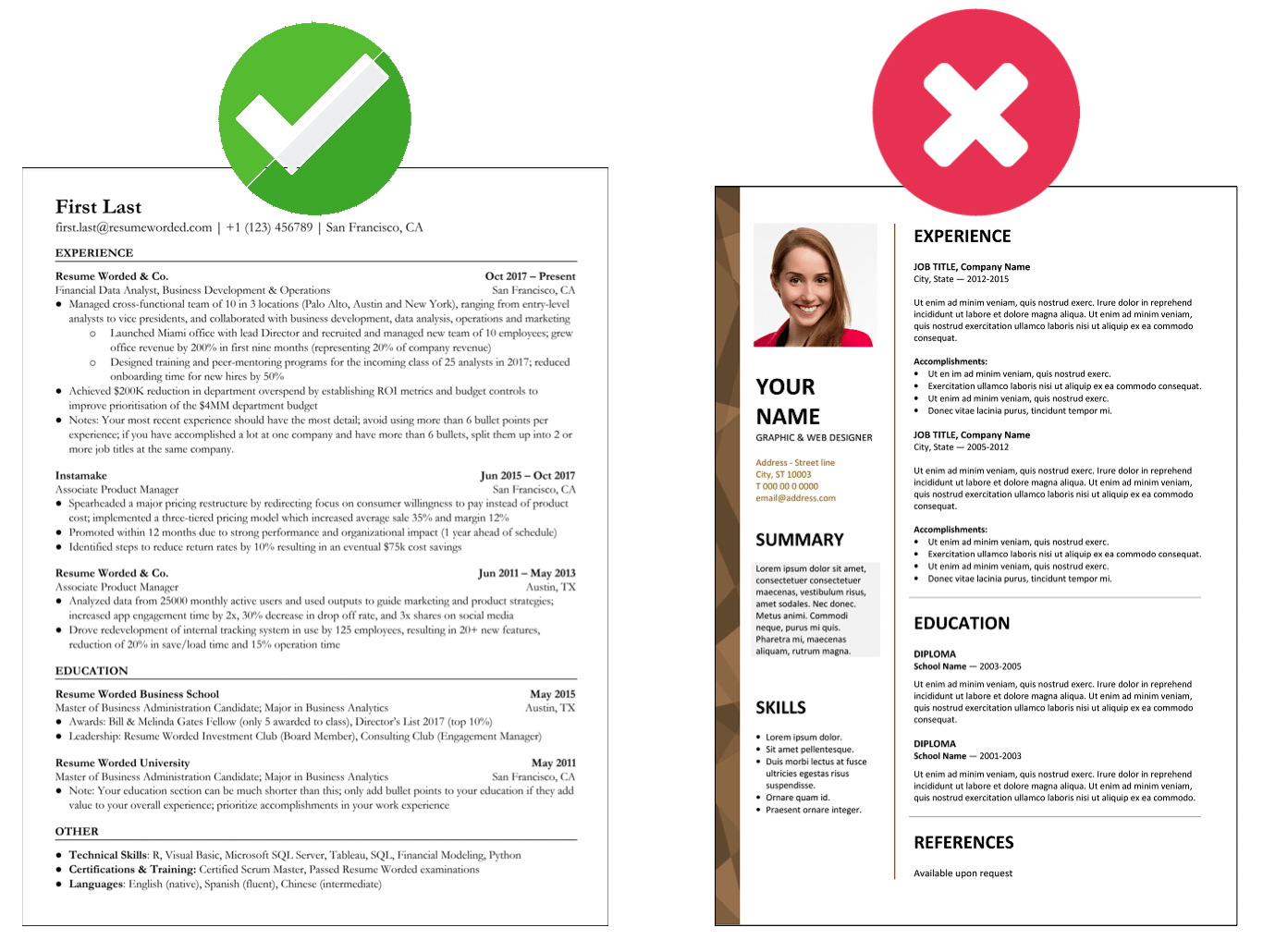
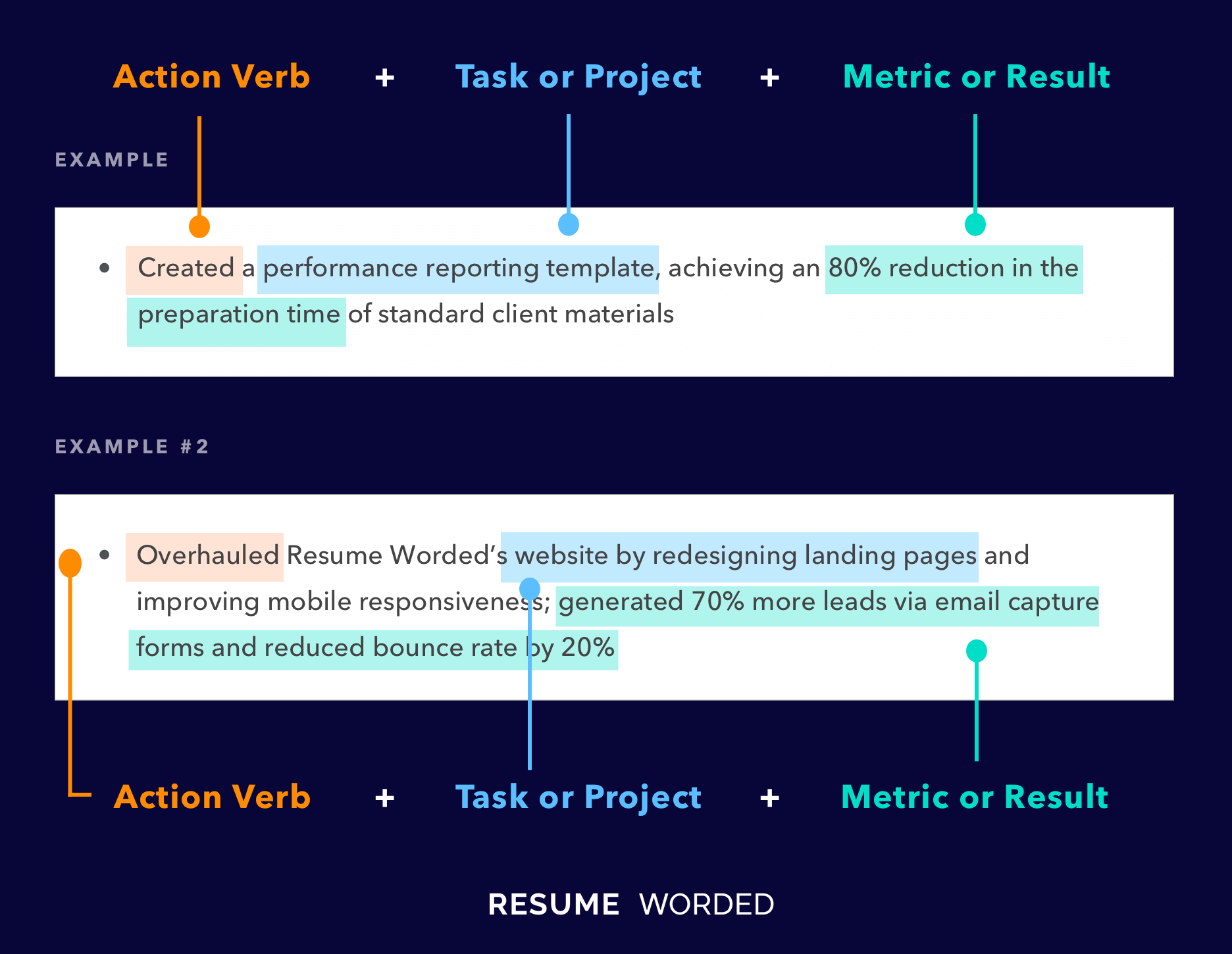
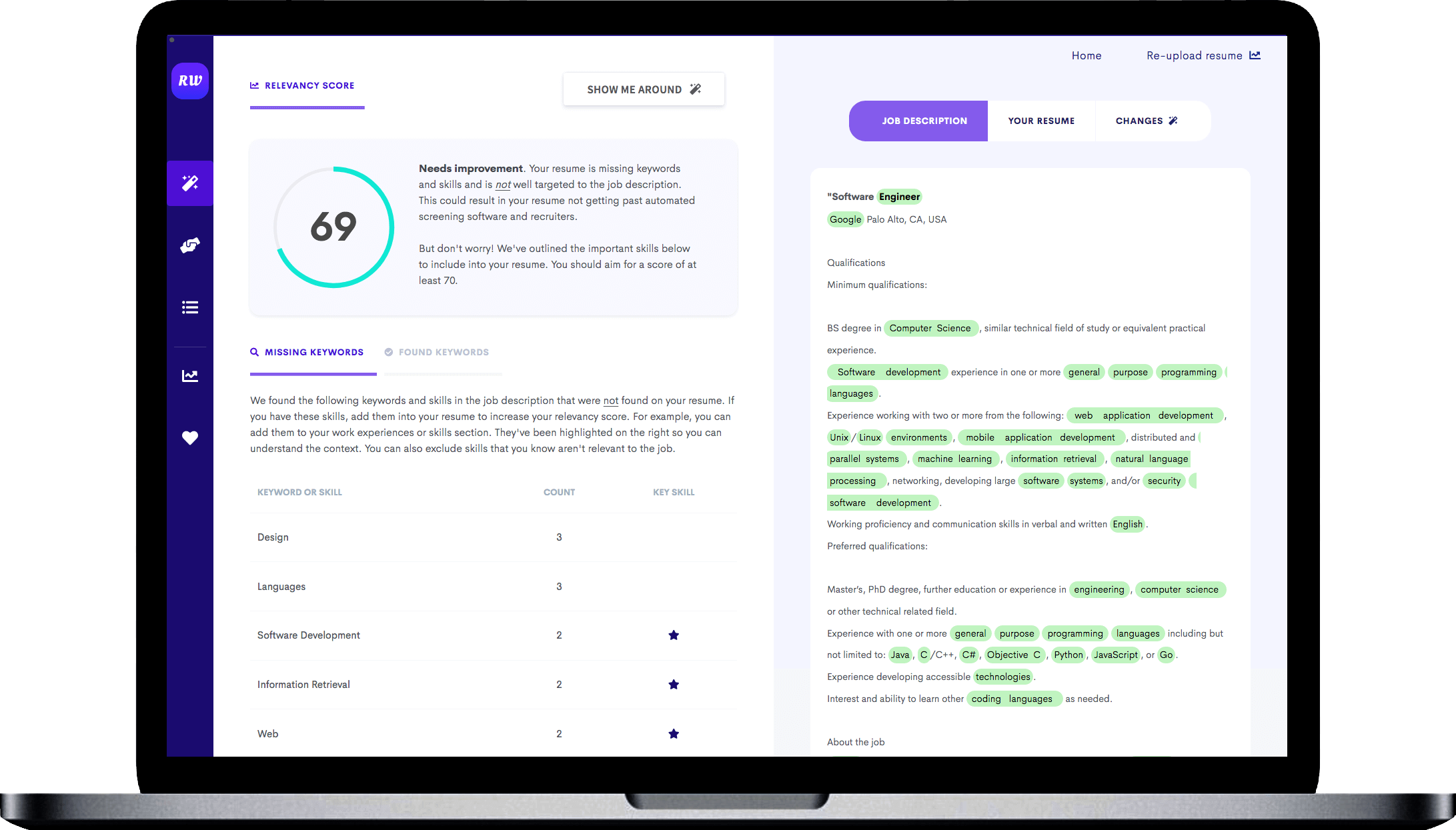
Showcase skills relevant to support C-levels
Different CEOs have different needs - for this position in particular, it’s important to read the job description carefully and even do some research of your own into the company’s CEO, given that their information is included. Make sure to include skills that tailor to the job posting. For example, if the executive travels often, emphasize your experience in making travel arrangements or serving as a travel assistant.